Since June 2015, our organizations have been working with the pulmonary arterial hypertension (PAH) and scleroderma communities to advocate for publicly funded access to all Health Canada-approved treatments for PAH, including Opsumit, Adempas, and Uptravi. We continue to be concerned about disparities that exist in the quality of care PAH patients may receive depending on what province they live in or whether they have private insurance.
Opsumit® (macitentan)
Approval:
In November 2013, Health Canada approved Opsumit for sale in Canada for the treatment of PAH. Opsumit, an endothelin receptor antagonist (ERA), is approved for the treatment of patients with idiopathic PAH (PAH with no known cause), heritable PAH, PAH associated with connective tissue disorders, and PAH associated with congenital heart disease. Opsumit is currently the only oral treatment proven in a randomized controlled trial to reduce long-term morbidity in patients, including reductions in PAH worsening and hospitalizations.
Public Reimbursement:
In October 2014, Quebec began providing public reimbursement for Opsumit. In January 2015, the national Common Drug Review (CDR) recommended that Opsumit be publicly funded (with criteria and a condition) for the long-term treatment of PAH to reduce morbidity in patients with Functional Class II or III. However, at the end of 2015, pricing negotiations for Opsumit through the pan-Canadian Pharmaceutical Alliance (pCPA) were closed. As a result, PAH patients outside of Quebec who could benefit from Opsumit, including those affected by SSc-PAH, were unable to access it through public funding.
In July 2019, pCPA reopened negotiations for Opsumit, and on December 23, 2020, a Letter of Intent (LOI) was signed. The last step in the reimbursement process is for each province to sign a Product Listing Agreement (PLA) with the manufacturer of Opsumit to make the treatment accessible to patients who can benefit from it.
As of February 1, 2023, Opsumit became accessible through public funding in all provinces except BC.
Advocacy Status:
In February 2021, PHA Canada contacted all provincial Ministers of Health and Public Drug Programs across the country urging them to provide swift reimbursement for Opsumit for PAH patients. In March 2021, PHA Canada launched a campaign of virtual meetings with provincial elected representatives across the country to ask that Opsumit be made publicly accessible as a treatment choice for all patients with PAH in Canada to allow for the best possible outcomes for those living with this disease.
If you are interested in speaking to your provincial elected representative about the importance of access to PAH treatments, please click here to contact us today.
Click here to download a copy of PHA Canada’s position paper highlighting the importance of access to all Health Canada-approved treatments for PAH, as well as the demonstrated benefits of Opsumit over other approved oral ERAs.
Uptravi® (selexipag)
 Approval:
Approval:
In January 2016, Health Canada approved Uptravi for sale in Canada for the treatment of PAH. Uptravi, the first oral selective IP prostacyclin receptor agonist, is approved for the long-term treatment of idiopathic PAH (PAH with no known cause), heritable PAH, PAH associated with connective tissue disorders, and PAH associated with congenital heart disease, to delay disease progression in adult patients with Functional Class II or III.
Public Reimbursement:
In October 2016 the Common Drug Review (CDR) recommended that Uptravi be publicly funded in the rest of Canada for the long-term treatment of PAH, in patients whose disease is not being adequately controlled with a first- and second-line therapy, but with a significant price reduction. In December 2017, the pan-Canadian Pharmaceutical Alliance (pCPA) successfully completed pricing negotiations for Uptravi.
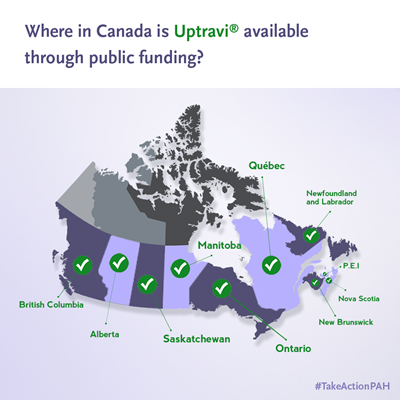
As of the end of 2018, Uptravi is accessible through public funding in every Canadian province.
Adempas® (riociguat)
Approval:
In March 2014, Health Canada approved Adempas for sale in Canada for the treatment of PAH. Adempas, a soluble guanylate cyclase stimulator, is also the only drug therapy approved for chronic thromboembolic pulmonary hypertension (CTEPH).
Public Reimbursement:
In December 2015, the Common Drug Review (CDR) recommended that Adempas be publicly funded (with criteria and a condition) for the treatment of PAH, as monotherapy or in combination with endothelin receptor antagonists (ERAs), in adult patients with Functional Class II or III. Quebec declined to list Adempas for PAH for public funding in February 2015, while Saskatchewan denied public funding in September 2016. In January 2017, it was announced that pricing negotiations for Adempas for PAH through the pan-Canadian Pharmaceutical Alliance (pCPA) had been closed because an agreement could not be reached, leaving patients throughout the country unable to access it through public funding.
Advocacy Status:
The PAH and scleroderma communities continue to urge governments to provide public reimbursement for Adempas for PAH.
PAH Advocacy and Reimbursement Timeline
Please click here for a detailed advocacy and reimbursement timeline.
Canada’s Drug Approval and Funding Process
Please click here for more information on Canada’s drug approval and funding process.